A recent study from Loughborough University demonstrates the impact of organosilane surface modifications on cardiovascular implants. The research focuses on modifying surfaces with various silane groups such as amine, methyl, and thiol to selectively enhance endothelial cell growth while suppressing smooth muscle cell proliferation. This dual effect helps address two critical issues in cardiovascular implants: improving endothelialization and preventing thrombosis.
Cardiac endothelial cells (ECs) and smooth muscle cells (SMCs) were investigated on surfaces treated with various organosilane groups, including amine, methyl, thiol, methacrylate, and fluorine. The results demonstrate that aminosilane-modified surfaces significantly enhance endothelial cell proliferation while reducing smooth muscle cell activity. This selective control is crucial for developing cardiovascular devices that promote healthy tissue integration while minimising clot formation.
The preparation of organosilane-modified glass coverslips
Borosilicate glass coverslips were first treated with oxygen plasma for 2 minutes (60 W Henniker Plasma HPT-200). Glass coverslips were pre-treated before being immersed in organosilane solutions. Plasma treatment activates the surface, ensuring strong bonding with the silane groups, which improves the bioactivity of the implants and promotes better cell interactions.
Modification Process Summary:
- Coverslips were cleaned with acetone, isopropanol, and methanol
- Plasma-treated coverslips were immersed in solutions containing selected organosilane chemistries:
- Methyl (-CH₃): Dimethyldichlorosilane (DCDMS)
- Amine (-NH₂): 3-Aminopropyltriethoxysilane (APTES)
- Thiol (-SH): 3-Mercaptopropyltrimethoxysilane (MPTS)
- Methacrylate (-CH₂=C-COOR): 3-(Trimethoxysilyl)propyl methacrylate (TMSPM)
- Fluorine (-F): 1H,1H,2H,2H-Perfluorooctyltriethoxysilane (CDTFS)
- Samples were thoroughly rinsed before cell culture.
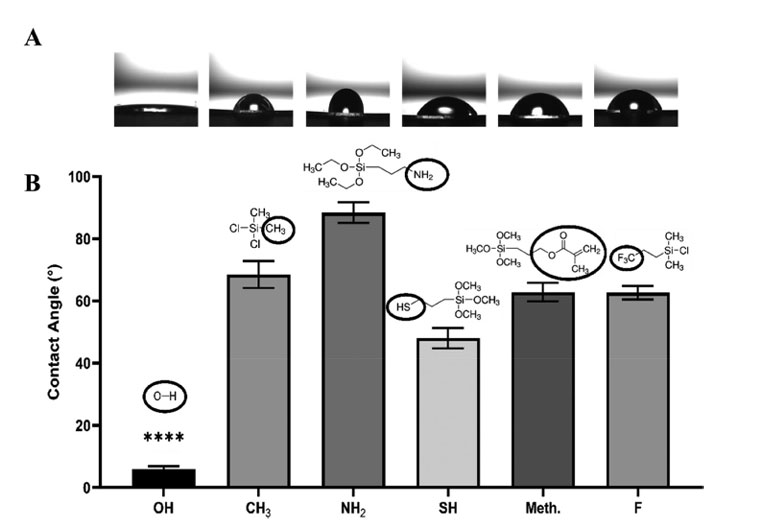
Results: Organosilane Modification Increases Water Contact Angle
Wettability experiments revealed that organosilane surface modification significantly increased surface hydrophobicity. As shown in the goniometry images (Figure 1A) and the plot (Figure 1B), the oxygen plasma-treated glass surface had a high wettability with a contact angle of approximately 5°. The introduction of organosilane functional groups resulted in a marked increase in contact angle, indicating a significant change in wettability compared to the plasma-treated hydroxyl control surfaces (p ≤ 0.0001).
[1] Figure 1: Courtesy of J Biomedical Materials Res - 2024 - Acar - Tuning Surface Chemistry Impacts on Cardiac Endothelial and Smooth Muscle Cell. Wettability analysis of organosilane-modified surfaces. (A) Static angle representative images, (B) results represented as the mean average contact angle values, N = 3, n = 20 with error bars representing ± SEM, p ≤ 0.0001.
The Bigger Picture
This work demonstrates the importance of precision surface modification in biomedical applications. Researchers achieved superior chemical bonding by employing plasma treatment for surface activation, enabling innovative strategies for cell-specific interactions on cardiovascular implants. This research demonstrates the potential of plasma-enhanced surface modification to advance safer, more effective medical devices.
Keywords
- Plasma Treatment
- Surface Modification
- Endothelialization
- Cardiovascular Implants
- Bioactivity
- Surface Chemistry
References
[1] Figure 1: Courtesy of J Biomedical Materials Res - 2024 - Acar - Tuning Surface Chemistry Impacts on Cardiac Endothelial and Smooth Muscle Cell. Wettability analysis of organosilane-modified surfaces. (A) Static angle representative images, (B) results represented as the mean average contact angle values, N = 3, n = 20 with error bars representing ± SEM, p ≤ 0.0001.
Join the Conversation
Readers are referred to the original paper accepted in November 2024, available through the provided DOI link, or click the link below for further details on the Henniker Plasma HPT-200.
Tuning Surface Chemistry Impacts on Cardiac Endothelial and Smooth Muscle Cell Development